RVINE, Calif.– Masimo (NASDAQ: MASI) announced today the findings of a study published in the Journal of Pain & Relief in which Dr. Jacques Chelly and colleagues at the University of Pittsburgh investigated the utility of Masimo Bridge®, an auricular field nerve stimulator, in reducing postoperative opioid requirements in patients undergoing kidney donor surgery.1 The researchers found that use of Bridge was associated with significant reductions in opioid use at 24 hours and pain at 24 and 48 hours after surgery, concluding that Bridge “may represent a complementary approach to minimize the postoperative requirement for opioid[s].”
Noting that opioids are still considered to be required after kidney donor surgery, in spite of their “established serious side effects including respiratory depression, postoperative nausea and vomiting” and that they “expose the donors to an unnecessary risk of opioid use disorder,” the researchers sought to evaluate whether the technique of percutaneous nerve field stimulation might provide benefit during the postoperative period. To evaluate this, they enrolled 20 patients undergoing kidney donor surgery, divided into two groups of 10. Both groups underwent the same enhanced recovery after surgery (ERAS) protocol based on a multimodal analgesic approach. The primary endpoint was opioid requirement, measured as oral morphine equivalent (OME), 24 hours after surgery. They also evaluated pain (on a 0 to 10 scale) 24 and 48 hours after surgery. In the experimental group (n=10), Masimo Bridge was fitted on patients in the post-anesthesia care unit.
The researchers found that, compared to the control group (n=10), the patients in the Bridge group had a 75.4% reduction in OME (8.3 ± 9.6 mg vs. 33.5 ± 37.3 mg, p = 0.03) and a 58.3% reduction in pain (2.5 ± 2.0 vs. 6 ± 1.4, p < 0.001) at 24 hours. At 48 hours, they had a 16.1% reduction in OME (22.4 ± 19.5 mg vs 26.7 ± 21.9 mg, p = 0.33) and a 73.3% reduction in pain (1.6 ± 1.6 vs. 6.0 ± 2.8, p = 0.0004). There was no difference in non-opioid analgesic use between the two groups. “Tolerability” of the Bridge device was reported as “excellent” by 78% of participants.
The researchers concluded, “This study suggests that the NSS-2 [Masimo] Bridge device may be of significant value in controlling postoperative opioid consumption and pain. This is especially interesting in the context of the current opioid epidemic and associated risk of opioid use disorder (OUD) in surgical patients. However, a prospective randomized placebo control design is required to confirm our findings.”
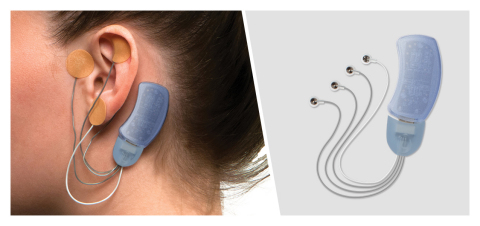
Masimo Bridge, the first evidence-based, drug-free, non-surgical device of its kind, has been cleared by the FDA for use in the treatment of clinical symptoms associated with opioid withdrawal, but is not currently FDA-cleared for postoperative pain management. The solution consists of a wearable, single-patient-use, percutaneous neurostimulator, fitted behind the ear, which applies gentle electrical impulses to branches of the cranial nerves around the ear. By using neuromodulation to aid in the reduction of withdrawal symptoms, Bridge may help patients with OUD successfully transition into a treatment program. In clinical testing, Bridge was found to reduce opioid withdrawal symptoms within 15-30 minutes and provide continuous relief for as long as it was applied, which can be up to 120 hours per device, allowing opioids to leave the body. In a study of 73 adults with OUD, it was shown that opioid withdrawal symptoms (such as increases in resting pulse rate, sweating, restlessness, bone or joint aches, tremors, and anxiety) were reduced by 85% after the first hour of using the device and 97% after 5 days of use (measured using the clinical opiate withdrawal scale).2
Study author Dr. Chelly commented, “Auriculotherapy is an ancient technique that has been used for centuries to treat pain. However, it requires long and specialized training and experience to be used effectively on patients. The Bridge device, by contrast, provides similar therapy but requires only limited training. Bridge offers the possibility of allowing many more patients to benefit from auriculotherapy, especially at a time when the use of opioids is so controversial.”
Joe Kiani, Founder and CEO of Masimo, said, “With FDA clearance, we have been marketing Bridge for opioid withdrawal for over a year and have seen amazing success stories. With this new promising study from UPMC, we will now work towards getting Bridge cleared for postoperative pain treatment worldwide. Last year, over 83,000 people died in the US alone from drug overdoses, with almost 63,000 of them killed by opioids, many of them from prescription opioids used post surgery.3 While we hope that with our FDA-pending, breakthrough-designated Masimo SafetyNet™ opioid solution, we will catch opioid overdoses early enough to save lives, it would be best if we can avoid the unnecessary use of opioids altogether. We hope additional studies will confirm that Bridge can help reduce the use of opioids.”