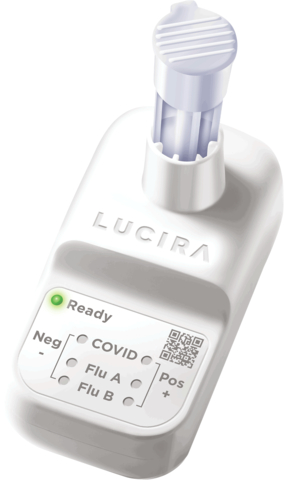
BURLINGTON, Ontario– KSL | Pulse Scientific, specializing in diagnostic, medical and laboratory products, announced today national distribution of the first and only over-the-counter combination test for COVID-19 and Flu, packaged and designed for self-testing at home. Developed by Lucira Health Inc., the test has been approved by Health Canada and is now available to institutions, health care providers, agencies and consumers in time for the fall and winter COVID and Flu season. The Lucira COVID-19 & Flu Test is a 99% accurate rapid molecular test that delivers results in 30 minutes or less from one shallow nasal swab.
“A good indicator of the coming flu season is often the recent season in Australia. Because Australia had an alarming spike in COVID and flu cases and we could see a substantial increase in North America. Many are projecting a ‘Twindemic’ of COVID and flu. Determining which infection someone has is difficult since flu and COVID present with similar symptoms. Lucira’s new product can be a game changer for medical staff and patients needing to determine the best course of action quickly,” says Kevin Lawson, President and CEO of KSL.
Lucira’s single-use test fits in the palm of a hand, runs on two AA batteries, and with one nasal swab, provides a positive or negative result for COVID-19, Flu A, and Flu B in 30 minutes or less. Each Lucira test is packaged with everything needed for the test. There is no separate reader or instrument to purchase and maintain.
“Lucira’s team did a great job developing this at-home test. We are ready to support the medical community and the public in Canada with ready access to fast and accurate testing,” add Lawson.
The Lucira testing platform is a Nucleic Acid Amplification Test (NAAT) with sensitivity and specificity comparable to lab-based PCR assays. Lucira’s standalone COVID-19 Test was the first at-home COVID-19 test to receive an EUA from the FDA in November 2020. The Lucira COVID-19 & Flu Test, which uses the same technology platform, was granted FDA EUA for point-of-care use earlier this month.
Health Canada has granted authorization under Interim Order No. 3. for emergency use and commercialization of this first and only at-home test for COVID & Flu. Health Canada’s decision is based on performance data including clinical trial results demonstrating comparable sensitivity and specificity to leading lab-based PCR assays.
The test also recently received CE marking, meeting EU health and safety requirements. Submissions to other regulatory authorities are in progress.