BERLIN, Germany– TME Pharma N.V. (Euronext Growth Paris: ALTME), a biotechnology company focused on developing novel therapies for treatment of cancer by targeting the tumor microenvironment (TME), announces a positive update on survival at 18 months for patients receiving NOX-A12 with the VEGF inhibitor bevacizumab and radiotherapy, and provides an overview on upcoming clinical development plans for NOX-A12 in the aggressive adult brain cancer, glioblastoma.
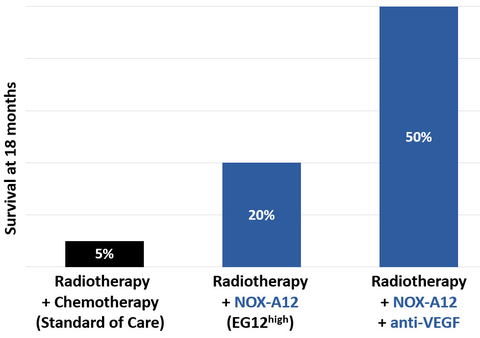
The percentage of patients who were alive 18 months after start of therapy of NOX-A12 with the VEGF inhibitor bevacizumab and radiotherapy is currently 50% (with the possibility to increase to 67% with the next patient reaching 18 months) which exceeds by 10-fold the 18-month survival of 5% observed in the matched group of patients receiving standard of care1. Since neither bevacizumab (anti-VEGF) alone, nor bevacizumab plus radiotherapy have previously shown to extend survival, the strong increase in survival can be attributed to the complementary mechanism of action of NOX-A12 with bevacizumab and radiotherapy2. The survival rate of the NOX-A12 triple combination also exceeds the 18-month survival of 20% seen in the patients with high levels of the NOX-A12 predictive biomarker EG12 receiving NOX-A12 + radiotherapy alone3, which further supports NOX-A12’s potential to synergize with VEGF inhibition in glioblastoma (see figure below).
The median overall survival has now reached 18 months and is expected to improve further as the remaining patients continue to receive treatment or follow-up care4. Two of the three living patients are clinically stable despite radiographic tumor progression at last report from treating clinicians, including the patient who achieved complete response, now completing 22 months therapy. As a reminder, the matched standard of care reference cohort achieved a median overall survival of 10.5 months.
The NOX-A12-based therapy has now delivered median overall survival exceeding all the relevant competitor studies conducted in the US or EU involving newly diagnosed, chemotherapy-resistant (MGMT unmethylated) glioblastoma patients despite recruiting more difficult to treat patients whose tumors could not be fully removed by surgery5. NOX-A12 in combination with bevacizumab and radiotherapy continues to show an excellent safety and tolerability profile similar to that noted in previous publications.
In the upcoming 6 months the key regulatory steps for NOX-A12 program in brain cancer will include the following:
- Q4 2023 – Request advice in October from US Food and Drug Administration (FDA) on next trial design and eligibility for expedited regulatory pathways, such as Fast-Track Designation. Feedback expected in late December.
- Q1 2024 – Submit IND6 application for glioblastoma with the US FDA along with expedited regulatory pathway access request. Successful IND filing and feedback targeted by end of Q1 2024.
TME Pharma plans to keep the market updated on the progress of these regulatory discussions. The goal is to have an FDA approved clinical trial protocol in glioblastoma with an expedited regulatory path by the beginning of April 2024 in order to secure the funding for the necessary clinical trial via partnership, investment or other strategic transaction types.
“We believe that survival data from the NOX-A12 bevacizumab expansion arm is now sufficiently mature and have made the decision to request advice from the US regulatory authority in the coming weeks. Once we have feedback from the FDA, we plan to submit an IND and request access to an expedited pathway for approval. We believe that having a clear path to marketing approval in brain cancer, validated by FDA, will significantly increase the attractiveness of NOX-A12 to investors and potential partners.” said Aram Mangasarian, CEO of TME Pharma. “While we believe that the FDA will request a randomized clinical trial examining two doses of NOX-A12 combined with bevacizumab and radiotherapy and to compare these two dose regimens against standard of care as a next step, they may also request that these two doses of NOX-A12 be tested only with radiotherapy (without bevacizumab) in order to quantify precisely the benefit of the combination with NOX-A12 and bevacizumab, which would add two additional arms to the clinical trial. We anticipate that an additional trial or an expansion of the upcoming trial would be required for full approval.”

