LOWELL, Mass. — RevBio, Inc. said the U.S. Food and Drug Administration has granted investigational device exemption approval to initiate a multicenter, randomized controlled pivotal clinical trial evaluating its regenerative bone adhesive, TETRANITE®, for cranial flap fixation following brain surgery.
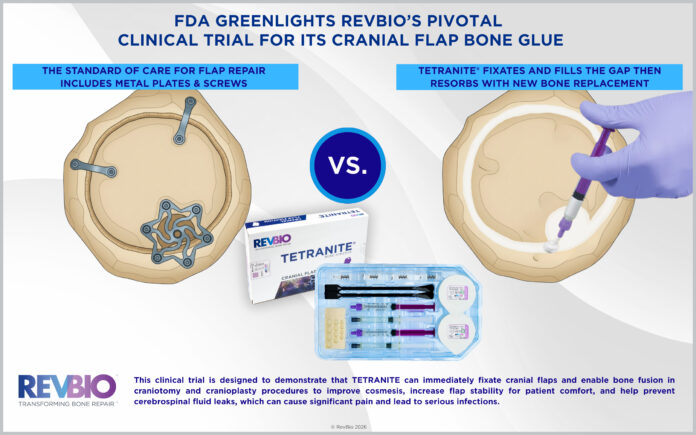
The approval allows RevBio to begin the final phase of clinical development ahead of potential commercial clearance. The study is designed to demonstrate that TETRANITE can immediately secure cranial bone flaps and promote bone fusion in craniotomy and cranioplasty procedures, with the goal of improving cosmetic outcomes, increasing flap stability for patient comfort, and reducing the risk of cerebrospinal fluid leaks that can lead to pain and serious infections.
“This novel regenerative adhesive has the potential to change how cranial restoration procedures are performed and significantly improve outcomes for patients,” said Timothy R. Smith, MD, PhD, MPH, director of the Computational Neuroscience Outcomes Center and a practicing neurosurgeon at Brigham and Women’s Hospital in Boston, Massachusetts.
The pivotal study, known as the T-RESTORE II trial, is expected to enroll up to 204 patients. Approximately half of the participants will receive TETRANITE, while the remaining patients will be treated using the current standard of care, which relies on titanium plates and screws to secure cranial bone flaps.
Craniotomy procedures are commonly performed to remove brain tumors, treat aneurysms, relieve intracranial pressure, or enable deep brain stimulation. During these surgeries, a section of the skull is temporarily removed, creating a continuous gap, or kerf line, between the bone flap and the surrounding skull. While plates and screws are used to reattach the flap, the kerf line is typically left unfilled, limiting bone integration because the gap is too wide for bone cells to cross.
As a regenerative bone adhesive, TETRANITE is applied directly into the kerf line, where it immediately fixates the bone flap, fills the gap, and creates a biological bridge that enables bone cells to migrate from the surrounding skull. This process is intended to support bone fusion and allow the flap to heal as a solid structure.
The FDA has approved up to 15 clinical sites to participate in the trial. RevBio said it is actively recruiting sites based on guidance from its neurosurgical advisory board. The agency’s decision was supported by results from a pivotal preclinical study, surgeon handling evaluations, and the company’s earlier pilot clinical trial, known as T-RESTORE I.
“This pivotal IDE trial allows us to conduct the final testing required for the commercial approval of our lead indication in the $10 billion market of targeted bone glue applications,” said Brian Hess, CEO and founder of RevBio, Inc. “The fact that RevBio now has two active pivotal stage programs highlights our ability to scale this technology as a platform.”