SEOUL, South Korea — South Korean biotech company Galux says it has reached a new benchmark in AI-driven antibody engineering, reporting that its design platform generated high-affinity therapeutic antibody candidates from a design pool of only 50 sequences per target epitope — a fraction of what is typically required in traditional discovery methods.
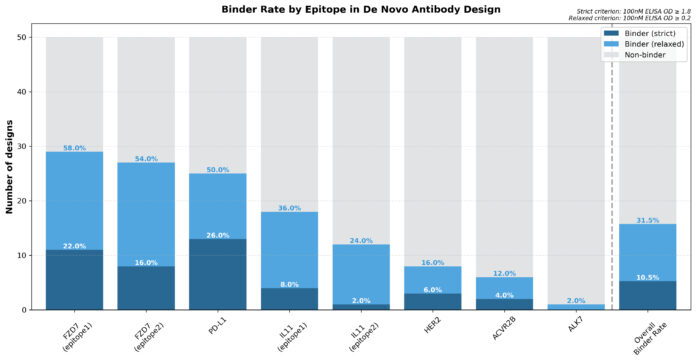
In new experimental data released by the company, Galux said its platform, GaluxDesign, produced an overall hit rate of 31.5 percent across eight epitopes. Roughly 10.5 percent of designs showed therapeutically meaningful affinity, including several antibodies that reached picomolar binding levels. All candidates were validated in full-length IgG format, suggesting they function as viable drug leads without added engineering steps.
Chaok Seok, CEO of Galux, said the results point to a shift from conventional library-based discovery toward more predictable design-driven development. “This performance represents more than a numerical improvement in success rate, it reflects a fundamental shift from discovering antibodies to rationally designing them,” Seok said, noting that traditional antibody discovery relies on large, probabilistic screening libraries and multiple rounds of affinity maturation.
Antibody development has historically required sifting through millions of variants to identify high-affinity binders, followed by extended humanization and optimization work. Galux argues that generating potent binders from only dozens of sequences — and doing so within weeks — marks a meaningful departure from those norms.
The data builds on earlier work from the company showing de novo antibody design across eight therapeutic targets, including PD-L1, HER2, EGFR S468R, ACVR2A/B, FZD7, ALK7, CD98hc, and IL-11. That effort yielded binders with affinities as strong as 9 picomolar. In the PD-L1 program, Galux resolved an antibody–antigen complex using cryo-EM with a 1.1 Å interface RMSD, supporting claims that the designs were structurally faithful to computational predictions.
The company also reported that functional traits — including specificity for mutational variants and subtypes — could be intentionally encoded during the design stage and retained in IgG format. Together, Galux says, the results demonstrate that structurally accurate and functionally tunable antibodies can now be produced from a relatively small set of AI-generated sequences.
As AI-based antibody discovery accelerates across the industry, Galux maintains that its platform distinguishes itself through validated precision across multiple metrics: structural accuracy, functional developability, target diversity, and small-design efficiency. Seok said the company now plans to extend the approach toward more complex antibody architectures and multi-target modalities.
“Our next step is to take this level of design control toward more advanced modalities, including multi-target binders and new functional architectures, and demonstrate how AI can expand the boundaries of what therapeutic antibodies can do,” he said.