LOS ANGELES, Calif. — Terray Therapeutics said it has introduced a new version of its EMMI Predict model, known as TerraBind, which the company says sets a new performance benchmark for AI-driven small molecule drug discovery by delivering higher accuracy while operating significantly faster than existing approaches.
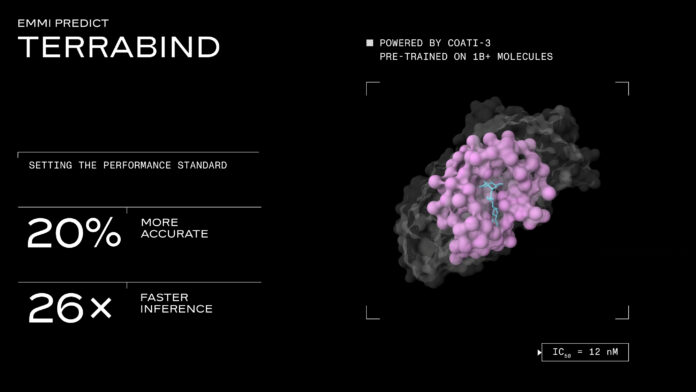
According to the company, TerraBind is a universal potency prediction model designed to operate at the scale required for modern small molecule drug development. Terray said the model achieves approximately 20 percent higher accuracy than leading open-source models while running up to 26 times faster, cutting inference costs by as much as 96 percent.
Terray’s broader platform, known as Experimentation Meets Machine Intelligence, or EMMI, is designed to integrate large-scale experimentation with machine learning to improve the speed, cost efficiency, and success rates of drug discovery programs. The platform underpins Terray’s internal discovery efforts as well as partnered programs with pharmaceutical companies including Bristol Myers Squibb, Gilead Sciences, and Odyssey Therapeutics.
At the core of the EMMI platform is a full stack of AI models that address multiple stages of molecule design. Terray said its system is built to generate millions of candidate molecular structures, predict their drug-like properties, and select optimal compounds for synthesis and experimental testing, all driven by a proprietary chemistry foundation model.
Terray developed TerraBind to address a key computational bottleneck in potency prediction. Traditional prediction models often rely on computationally intensive diffusion-based approaches that make it impractical to evaluate millions of molecules at scale. By contrast, TerraBind was designed to operate efficiently across very large chemical libraries.
The company said the architectural shift also improves predictive accuracy. In side-by-side testing, TerraBind outperformed Boltz-2, a recently released comparison model, on both public benchmarks and internal drug discovery programs. On the CASP16 benchmark, TerraBind achieved a 16 percent higher Pearson correlation. Across 18 internal drug discovery targets spanning multiple protein families and chemical scaffolds, the model showed an average accuracy improvement of about 20 percent. Terray added that TerraBind matched Boltz-2 in ligand pose accuracy on benchmarks such as FoldBench, PoseBusters, and Runs N’ Poses, while delivering results far more quickly.
“This breakthrough in binding affinity prediction is a cornerstone in our AI-driven, experimental workflow,” said Jacob Berlin, Chief Executive Officer of Terray. “EMMI is grounded in the largest chemistry data set in the world, currently at 14 billion unique target-molecule measurements, which has consistently enabled us to find starting points for undruggable targets in the dark areas of chemical space.”
Terray attributed TerraBind’s performance to a rethinking of how binding predictions are generated. Rather than modeling full all-atom protein-ligand structures through diffusion processes, the model uses coarse-grained structural representations, focusing on protein residue centers and ligand heavy atoms. The company said this approach retains the information needed to predict binding affinity and binding location while eliminating unnecessary computational overhead.
TerraBind also serves as the foundation for Terray’s EMMI Select models, which are designed to identify optimal molecules for synthesis and testing during each discovery cycle. The company recently announced that these selection models incorporate uncertainty-aware techniques to improve decision-making, delivering time and cost savings of up to threefold in retrospective potency optimization studies.
Terray said the integrated EMMI platform enables it to combine internal and external data sources to compress drug discovery timelines, identify novel molecular scaffolds across a broad range of targets, and rapidly optimize new compounds. The company believes this approach represents a shift in how small molecule drugs are discovered and developed, with the potential to accelerate the delivery of new medicines through both internal and partnered programs.

